Platelet activation plays a key role in thrombus formation in cardiovascular pathologies (i.e. stenosis) and blood recirculating devices (i.e. prosthetic heart valves, artificial hearts, and ventricular assist devices). These pathologies and devices are characterized by regions of high shear stress with brief exposure times. While shear-induced platelet activation has been studied extensively, its link with the dynamic flow environment found in such pathologies and devices is poorly understood.
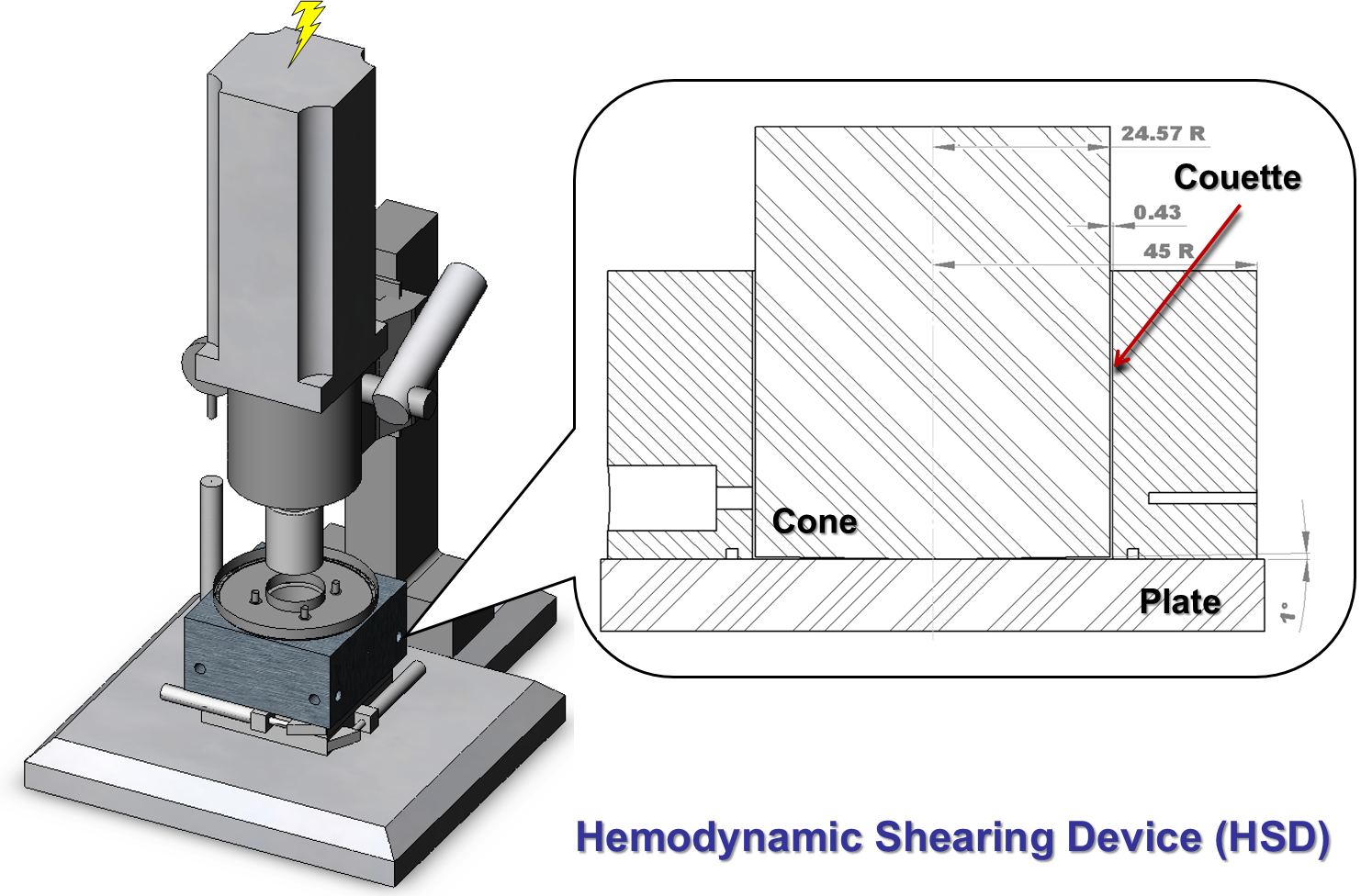
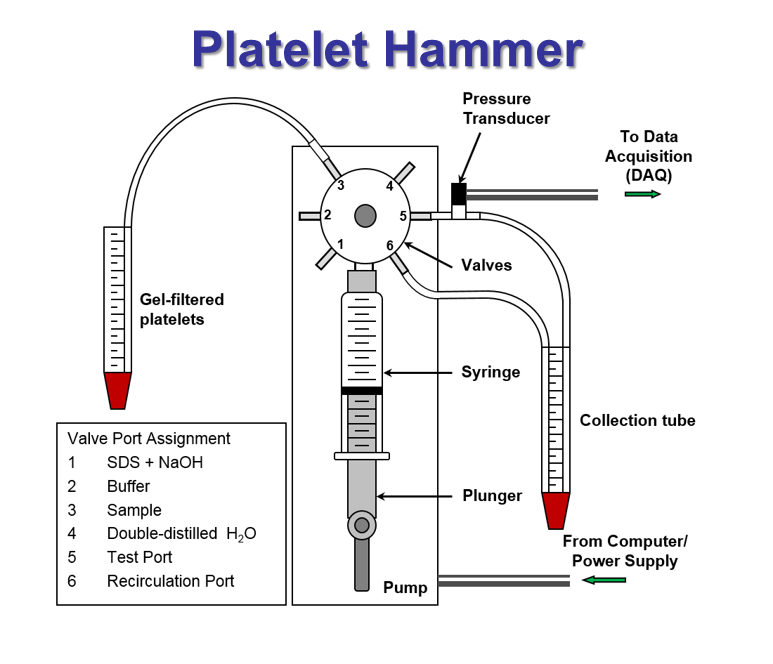
Our group uses several experimental and mathematical tools to study shear-induced platelet activation. The Hemodynamic Shearing Device (HSD) is a computer-controlled cone-plate-Couette viscometer that uniformly exposes platelets and other blood components to programmed dynamic high shear stress waveforms. The platelet hammer repeatedly subjects platelets to hypershear conditions, up to 4000 dyne/cm2, with exposure times on the order of milliseconds. A variety of techniques are used to measure the platelet and blood response. These methods include flow cytometry, differential interference contrast (DIC) microscopy, scanning electron microscopy (SEM), and chromogenic assays for lysis and platelet activation.
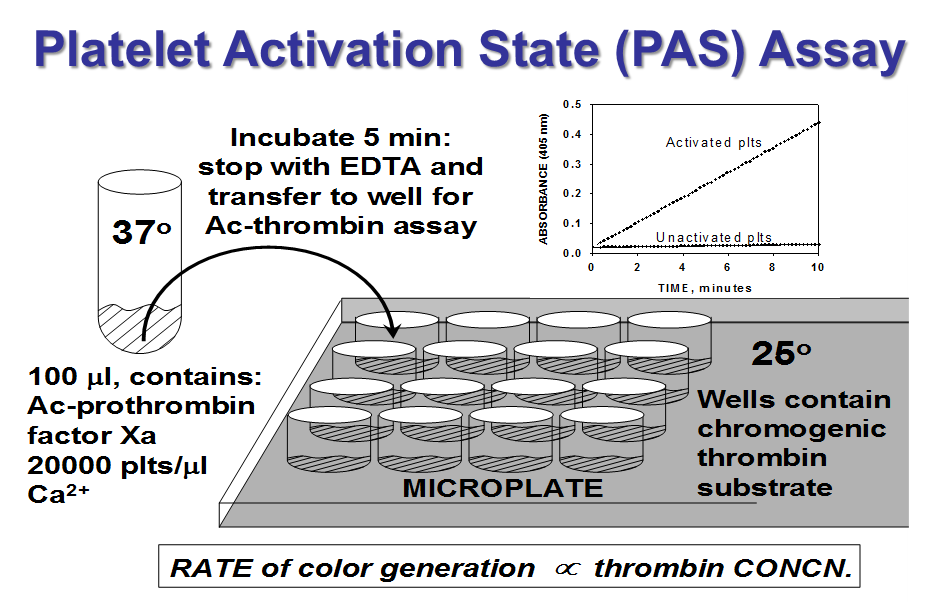
The Platelet Activation State (PAS) Assay
Platelet activation is measured using a variety of methods, but the quantification of thrombin generation provides valuable information about the role of platelets in the common pathway of the coagulation cascade. Thrombin is a potent agonist that activates and recruits other platelets to growing aggregates and clots. In the PAS assay, an acetylated version of prothrombin, the precursor to thrombin, is used. The resulting modified thrombin is unable to participate in the formation of insoluble fibrin (the clot material) or the positive feedback loop to further activate platelets. Thus, the modified thrombin provides an excellent marker of platelet activation when coupled with a chromogenic substrate, and the assay gives a one-to-one relationship with the applied agonist, shear stress.
Shear-Induced Platelet Sensitization
Patients implanted with prosthetic cardiovascular devices have an elevated risk of thromboembolism, despite antiplatelet and anticoagulant therapy. We hypothesized that this is due to continuing platelet activation subsequent to the initial high shear insult in the device region. Our group has performed several experiments to examine this behavior, and these have included measurements for platelet activation, aggregation, lysis, and treatment with several platelet agonists and inhibitors.
Antiplatelet Efficacy under Shear Stress
The efficacy of antiplatelet drugs under shear stress conditions prevalent in blood recirculating devices is not well understood. Pharmacological therapy in such cases is a trade-off between limiting thromboembolic complications and preventing bleeding incidents due to overdosing. We analyze the platelet activation after treatment with several antiplatelet drugs, including aspirin and dipyridamole, and exposure to dynamic shear stress conditions.
Platelet Activation and Blood Damage Models
Several groups have developed mathematical models to link blood damage to the shear stress conditions to which blood elements are exposed. However, few studies have extended this examination to platelets, and existing models tend to be limited in the range of applicable shear stress conditions and dynamicity. Our group has analyzed or developed several models using various mathematical approaches, while accounting for the past shear stress history, dynamic shear conditions, and behaviors such as shear-induced platelet sensitization.